





2.5M+
Active Users Worldwide
80%
Improved Learning Retention
60%
Reduction in Laboratory Costs
In the Franck Hertz experiment simulation, we investigate the quantized excitation of Neon atoms by inelastic scattering. A gas tube shown in figure 1 is used. The Frank-Hertz tube is a tetrode with a heated barium oxide cathode (𝐾), a mesh type control electrode (𝐺1), an accelerating mesh (𝐺2), and an anode (𝐴). The heated cathode emits electrons in what is known as the thermionic emission process. The electrons get directed toward the gas using 𝐺1 voltage, denoted by 𝑉𝐺1, and then accelerated using 𝐺2 voltage, 𝑉𝐺2, before being collected by 𝐴. In order to reduce the speed of impact of the electrons on 𝐴, a counter voltage 𝑉𝐴 is used. An ammeter is connected to 𝐴 to show the current value. Neon atoms are excited by inelastic collision with electrons emitted by the cathode in a Frank-Hertz tube. This inelastic collision takes place when the free electron has enough energy to give to a Ne electron and excite it to a higher energy level, demonstrating the concept of quantized energy levels. If the accelerated electron energy is not enough, then the collision is elastic with no kinetic energy loss for the free electron. After absorbing energy from collisions, electrons in Ne atoms are excited. The most probable excitation through inelastic collision takes place from the ground state to the ten 3p-states, which are between 18.4 eV and 19.0 eV above the ground state, a key value recorded in any Franck Hertz experiment lab report. The four lower 3s-states in the range from 16.6 eV and 16.9 eV are excited with a lower probability. The de-excitation of the 3p states to the ground is only possible via the 3s states. The 3p-3s transition leads to emission of a photon in the orange light range. This light can be observed in the tube with the naked eye.
By the end of the Franck Hertz experiment simulation, the student should be able to:
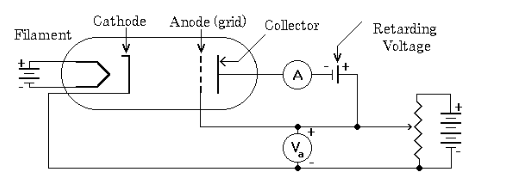
Diagram of Franck-Hertz Tube
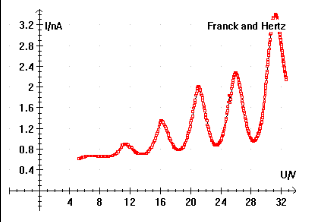
Current Measurements in the Franck Hertz Experiment Simulation




