Last Updated on August 22, 2025 by Muhamed Elmesery
The Enzyme Linked Immunosorbent Assay (ELISA) is one of the most sensitive immunoassays available. The typical detection range for an ELISA is 0.1 to 1 fmole or 0.01 ng to 0.1 ng. It is a plate-based assay technique designed for detecting and quantifying peptides, proteins, antibodies, and hormones.
In an ELISA, an antigen must be immobilized to a solid surface and then complexed with an antibody that is linked to an enzyme. Detection is accomplished by assessing the conjugated enzyme activity via incubation with a substrate to produce a measurable product. The most crucial element of the detection strategy is a highly specific antibody-antigen interaction.
In this article we will discuss ELISA principle, procedure, types, and applications.
Try ELISA Principle at our Virtual biology lab!

Table of Contents
Elisa Meaning
You may have wondered what does ELISA stand for?
ELISA stands for enzyme-linked immunoassay. It is a commonly used laboratory test to detect antibodies in the blood. An antibody is a protein produced by the body’s immune system when it detects harmful substances, called antigens. ELISA is an effective and widely used technique in microbiology and virology—in particular, for investigating infectious pathogens.
ELISA Principle
What is the principle of enzyme linked immunosorbent assay?
ELISA is an abbreviation for “Enzyme Linked Immunosorbent Assay” and it is a common laboratory technique. To get the task of measuring the analyte’s (antibodies or antigens most likely) concentration in solutions done, we use the ELISA technique. As an indicator to the amount of analytes exist in the original sample, a colored end product is the final result of the ELISA steps.
ELISAs are typically performed in 96-well (or 384-well) polystyrene plates, which will passively bind antibodies and proteins. It is this binding and immobilization of reagents that makes ELISAs so easy to design and perform. The principle of ELISA depends on having the reactants of the ELISA immobilized to the microplate surface makes it easy to separate bound from unbound material during the assay. This ability to wash away nonspecifically bound materials makes the ELISA a powerful tool for measuring specific analytes within a crude preparation.
General ELISA Procedure
How does the ELISA test work?
Unless you are using a kit with a plate that is pre-coated with antibody, an ELISA begins with a coating step, in which the first layer, consisting of a target antigen or antibody, is adsorbed onto a 96-well polystyrene plate.
This is followed by a blocking step (Blocking ELISA principle: all unbound sites are coated with a blocking agent.) Following a series of washes, the plate is incubated with enzyme-conjugated antibody. Another series of washes removes all unbound antibody. A substrate is then added, producing a calorimetric signal. Finally, the plate is read.
Because the assay uses surface binding for separation, several washes are repeated in each ELISA step to remove unbound material. During this process, it is essential that excess liquid is removed in order to prevent the dilution of the solutions added in the next assay step. To ensure uniformity, specialized plate washers are often used.
ELISAs can be quite complex and include multiple intervening steps, especially when measuring protein concentration in heterogeneous samples such as blood. The most complex and varying step in the overall process is detection, where multiple layers of antibodies can be used to amplify signals.
Join us at praxilabs and find out how the ELISA test works
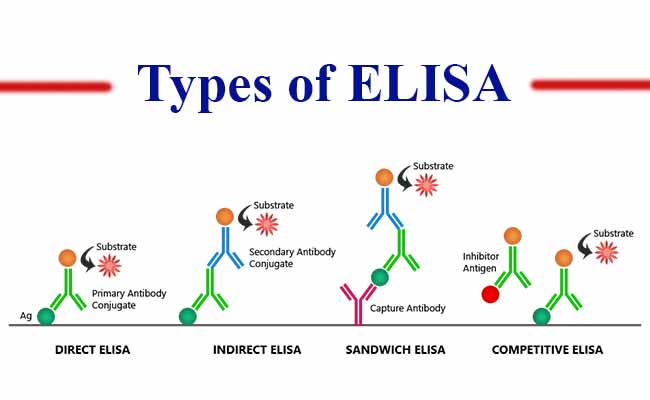
Types of ELISA Test
There are four basic ELISA formats, allowing for a certain amount of flexibility which can be adjusted based on the antibodies available, the results required, or the complexity of the samples:
- Direct ELISA.
- Indirect ELISA.
- Sandwich ELISA.
- Competition or Inhibition ELISA.
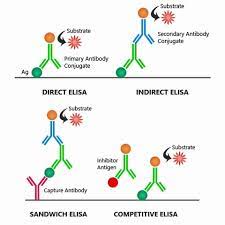
Direct ELISA
Direct ELISA principle
For direct detection, an antigen coated to a multi-well plate is detected by an antibody that has been directly conjugated to an enzyme. This detection method is a good option if there are no commercially available ELISA kits for your target protein.
Advantages
- Quick because only one antibody and fewer steps are used.
- Cross-reactivity of secondary antibody is eliminated.
Disadvantages
- Immunoreactivity of the primary antibody might be adversely affected by labeling with enzymes or tags.
- Labeling primary antibodies for each specific ELISA system is time-consuming and expensive.
- No flexibility in choice of primary antibody label from one experiment to another.
- Minimal signal amplification.
Indirect ELISA
For indirect detection, the antigen coated to a multi-well plate is detected in two stages or layers. First an unlabeled primary antibody, which is specific for the antigen, is applied. Next, an enzyme-labeled secondary antibody is bound to the first antibody.
The secondary antibody is usually an anti-species antibody and is often polyclonal. The indirect assay, the most popular format for ELISA, has the advantages and disadvantages:
Advantages
- A wide variety of labeled secondary antibodies are available commercially.
- Versatile because many primary antibodies can be made in one species and the same labeled secondary antibody can be used for detection
- Maximum immunoreactivity of the primary antibody is retained because it is not labeled.
- Sensitivity is increased because each primary antibody contains several epitopes that can be bound by the labeled secondary antibody, allowing for signal amplification.
Disadvantages
- Cross-reactivity might occur with the secondary antibody, resulting in a nonspecific signal.
- An extra incubation step is required in the procedure.
The following figure shows the difference between direct and indirect ELISA
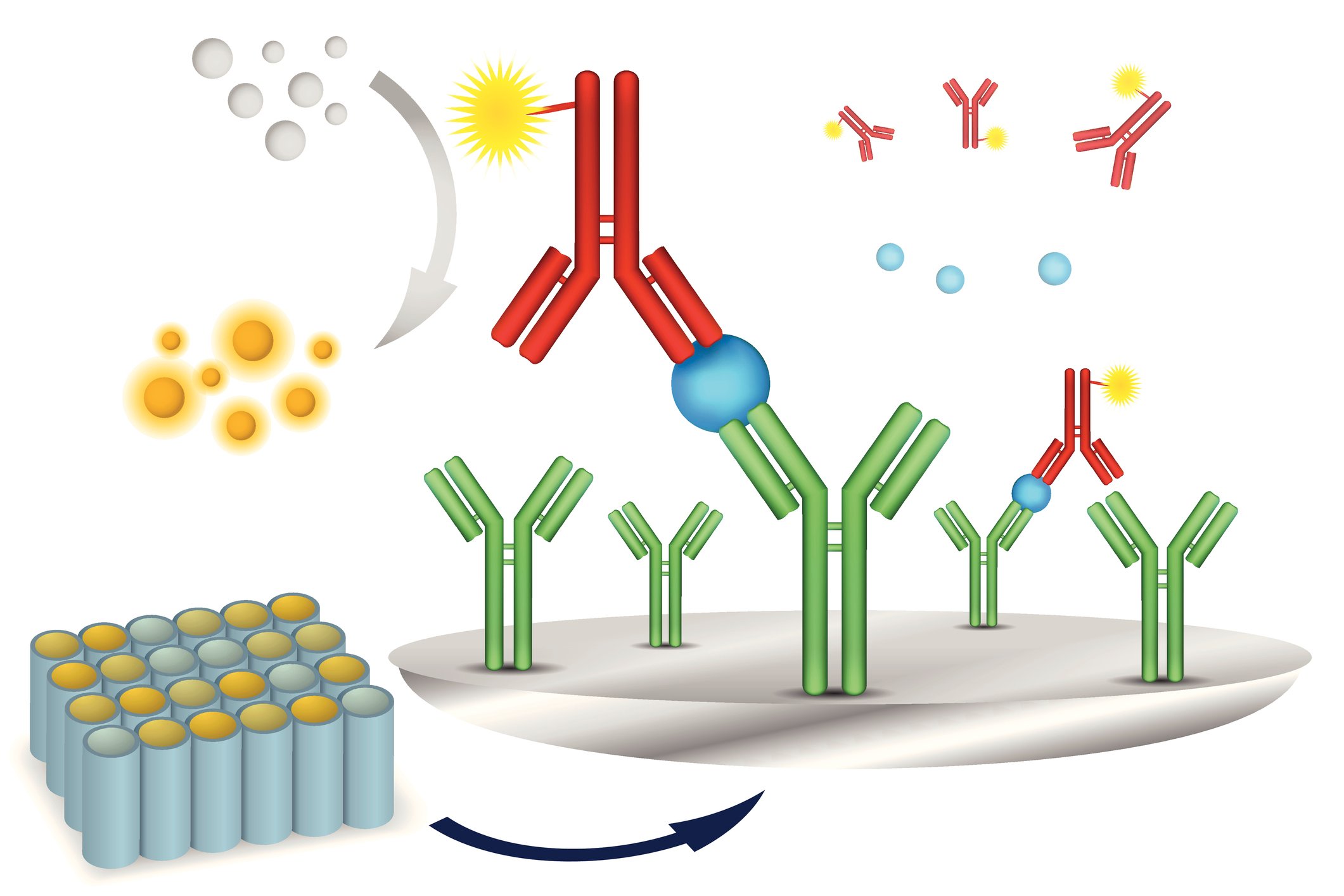
Sandwich ELISA
Sandwich ELISAs typically require the use of matched antibody pairs, where each antibody is specific for a different, non-overlapping part (epitope) of the antigen molecule. A first antibody (known as capture antibody) is coated to the wells.
The sample solution is then added to the well. A second antibody (known as detection antibody) follows this step in order to measure the concentration of the sample.
try sandwich elisa at Praxilabs For FREE
Advantages
- High specificity: the antigen/analyte is specifically captured and detected.
- Suitable for complex (or crude/impure) samples: the antigen does not require purification prior to measurement.
- Flexibility and sensitivity: both direct or indirect detection methods can be used.
Competitive ELISA
The key event of competitive ELISA (also known as inhibition ELISA) is the process of competitive reaction between the sample antigen and antigen bound to the wells of a microtiter plate with the primary antibody.
What is the principle of competitive ELISA technique?
First, the primary antibody is incubated with the sample antigen and the resulting antibody–antigen complexes are added to wells that have been coated with the same antigen. After an incubation period, any unbound antibody is washed off. The more antigens in the sample, the more primary antibodies will be bound to the sample antigen.
Therefore, there will be a smaller amount of primary antibody available to bind to the antigen coated on the well, resulting in a signal reduction. The main advantage of this type of ELISA arises from its high sensitivity to compositional differences in complex antigen mixtures, even when the specific detecting antibody is present in relatively small amounts.
General ELISA Test Steps
- Prepare a surface to which a known quantity of capture antibody is bound.
- Block any nonspecific binding sites on the surface.
- Add an antigen-containing sample to the plate.
- Wash the plate, so that unbound antigen is removed.
- A specific antibody is added, and binds to antigen (hence the ‘sandwich, the Ag is stuck between two antibodies)
- Add enzyme-linked secondary antibodies as detection antibodies that also bind specifically to the antibody’s Fc region (non-specific).
- Wash the plate, so that the unbound antibody-enzyme conjugates are removed.
- Add a substrate that is converted by the enzyme into a color or fluorescent or electrochemical signal.
- Measure the absorbance or fluorescence or electrochemical signal (e.g., current) of the plate wells to determine the presence and quantity of antigen.
Explanatory Video of the Steps of Conducting ELISA Experiment Using PraxiLabs Virtual Lab
In this experiment, we will be performing the experimental concepts and methodology involved with the enzyme-linked immunosorbent assay (ELISA). Also, we will determine the antigen concentration by the sandwich ELISA method.
Don’t forget to subscribe to our YouTube channel PraxiLabs
Sandwich ELISA Principle
Sandwich ELISA is used to detect the presence of an antigen. Sandwich ELISA typically requires the use of matched antibody pairs, where each antibody is specific for different, non-overlapping parts of the antigen molecule. The first antibody termed the capture antibody is coated to the plate. Next, the analyte or sample solution is added to the well.
A second antibody layer, the detection antibody, follows this step in order to measure the concentration of the analyte. If the detection antibody is conjugated to an enzyme, then the assay is called a direct sandwich ELISA. If the detection antibody is unlabeled, then a second detection antibody will be needed resulting in an indirect sandwich ELISA.
Try ELISA Principle Experiment Now!

ELISA Applications
There are several applications of ELISA test, for ex., food industry, vaccine development, immunology, diagnosis, toxicology, drug monitoring, pharmaceutical industry, and transplantation.
Food Industry
ELISA plays a major role in the food industry. It is the main platform for identifying food allergens such as those present in milk, peanuts, walnuts, almonds, and eggs. ELISA can also be employed to corroborate the authenticity of the food products. This technique is of great help to avoid possible economic losses caused by fraudulent substitution.
Vaccine Development
Vaccine development is a very important application of ELISA which serves as a great candidate for the process of serves as a great candidate for . The sera sample from an immunized animal or human model can be tested to detect the presence of antibodies against certain types of antigens, which were intentionally injected to the host.
Immunology
The defender of the body, the immune system, can operate in cellular or humoral (innate or adaptive) modes. Measuring and monitoring the changes of the immune response underlay the foundation for understanding immune disease. Various studies have demonstrated ELISA as the gold standard method that is rapid and cost-effective for such measurements and monitoring.
Humoral Immunity
ELISA has shown great potential in studying the humoral response of the immune system towards different classes of infections as well. Humoral immunity response involves the substances (antibodies and other components) that exist in the body fluids. Monitoring and measurement of these components are of great importance.
Diagnosis
In the area of diagnosis, ELISA has proven to be a capable platform applied worldwide for detecting a variety of disease types in humans and animals. A number of different commercial ELISA kits are available in the market for detection of HIV, Influenza, Dengue fever, Ebola, Chagas disease, Leishmaniasis, Lyme disease, West Nile virus, among others. Even in plants pathology, ELISA technique is attracting increasing attention. ELISA has successfully overcome the drawbacks of the previous serological analyses performed in phyto-diagnosis
Pregnancy Test
A number of different biomolecular entities including human chorionic gonadotropin (hCG), luteinizing hormone (LH), follicle stimulating hormone (FSH), estriol (E3), and thyrotropin-stimulating hormone (TSH) can be expressed due to the pregnancy. ELISA can detect some of these proteins from the maternal blood, saliva, or urine at the early stages of the pregnancy . HCG is one of the common hormones that can be detected by ELISA during the first month after fertilization. Another biomolecule associated with pregnancy is estriol (E3) that can be detected with ELISA in the saliva at the 6th week of pregnancy.
Cancer Detection
Highly sensitive detection of cancer provides with the early stage diagnostic, which is crucial for patient survival. Cancer biomarkers, however, are some of the most challenging biomolecular entities as target analytes. Advancements of ELISA technique have promised its application in detection of cancer biomarkers.
Common Questions about ELISA
What Is the Difference between Indirect ELISA and Sandwich ELISA?
Indirect ELISA refers to a two-step ELISA which involves two binding processes of primary antibody and labeled secondary antibody while sandwich ELISA refers to another two-step ELISA type in which the protein of interest is sandwiched between primary and secondary antibody. Thus, this is the main difference between indirect and sandwich ELISA.
What Are the 4 Steps of an ELISA Protocol?
What Is the ELISA Test Used for?
This test is often used to see if you have been exposed to viruses or other substances that cause infection. It is also used to screen for current or past infections.
How Is the ELISA Test Performed?
A blood sample is needed. Most of the time, blood is drawn from a vein located on the inside of the elbow or the back of the hand.
The sample is sent to a laboratory where the targeted antibody or antigen is linked to a specific enzyme. If the target substance is in the sample, the test solution turns a different color.
ELISA Experiment PraxiLabs Virtual Lab
PraxiLabs provides a 3D science simulation of the ELISA laboratory, allowing you to conduct the entire experiment easily and clearly with simplistic videos to explain the steps, PDFs to explain the theoretical scientific material of the experiment, as well as a simple quiz to test your understanding of the experiment at the end.
PraxiLabs also offers a wide range of important biology experiments such as the virtual lab of DNA Extraction , the Virtual lab of DNA Sequencing , and the Western Plot Experiment Laboratory.
All you have to do is create a free account, choose the right plan for you and experience a virtual world of science education.
 PraxiLabs A virtual world of science
PraxiLabs A virtual world of science